Check out this nifty carbon-catching "artificial leaf" system
Researchers from the University of Chicago Illinois have created an "artificial leaf" system capable of capturing 100 times more carbon than other tech.
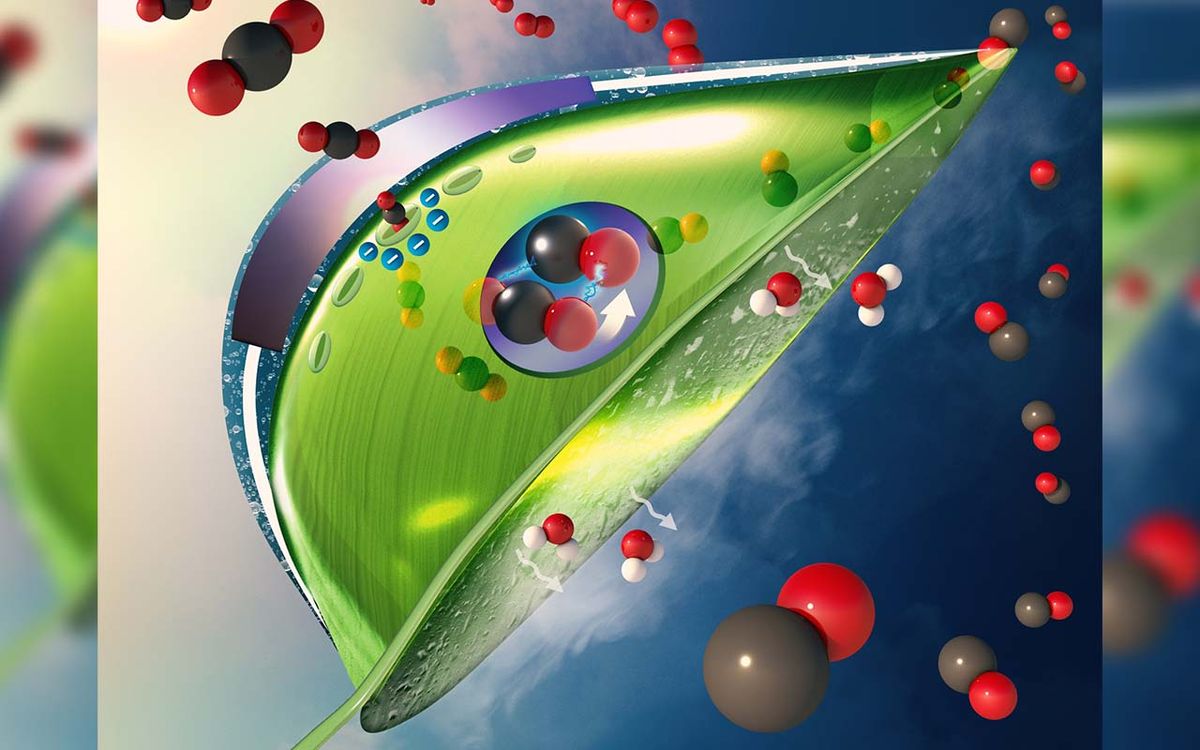
Climate change is a topic at the forefront of many minds around the world – especially following one of the hottest years recorded. Reducing our carbon footprint is a common focus.
Researchers from the University of Illinois Chicago have developed a system modelled after the process of photosynthesis that can capture carbon dioxide 100 times more effectively than any other current technology.
Pretty neat, eh?
It's not a literal leaf, as the "artificial leaf" label may suggest, but a system housed within an electrically-charged resin membrane with wet and dry compartments.
How it works is an organic solvent and carbon dioxide combine to create a bicarbonate byproduct, which is drawn into the wet compartment to dissolve into a reduced concentration of carbon dioxide suitable for use as fuel.
The whole thing is small enough to fit in a backpack, too.
I won't pretend to fully understand the science behind it – that's what the folks at New Atlas are for – but the fact the concept is now proven to work outside of a lab setting sounds extremely promising.
This part from the University of Chicago Illinois' official release is also impressive:
"When they tested the system, which is small enough to fit in a backpack, the UIC scientists found that it had a very high flux – a rate of carbon capture compared with the surface area required for the reactions – of 3.3 millimoles per hour per 4 square centimetres. This is more than 100 times better than other systems, even though only a moderate amount of electricity (0.4 KJ/hour) was needed to power the reaction, less than the amount of energy needed for a 1 watt LED lightbulb. They calculated the cost at US$145 per ton of carbon dioxide, which is in line with recommendations from the Department of Energy that cost should not exceed around US$200 per ton."University of Chicago Illinois
So, it's effective, cheap enough for wide use, and has the backing of science. Sounds good to me.
You can check out the full paper published in the Energy in Environmental Science journal, titled "Migration-assisted, moisture gradient process for ultrafast, continuous CO2 capture from dilute sources at ambient conditions".
It's a mouthful of a title, but who am I to argue with the results?




